
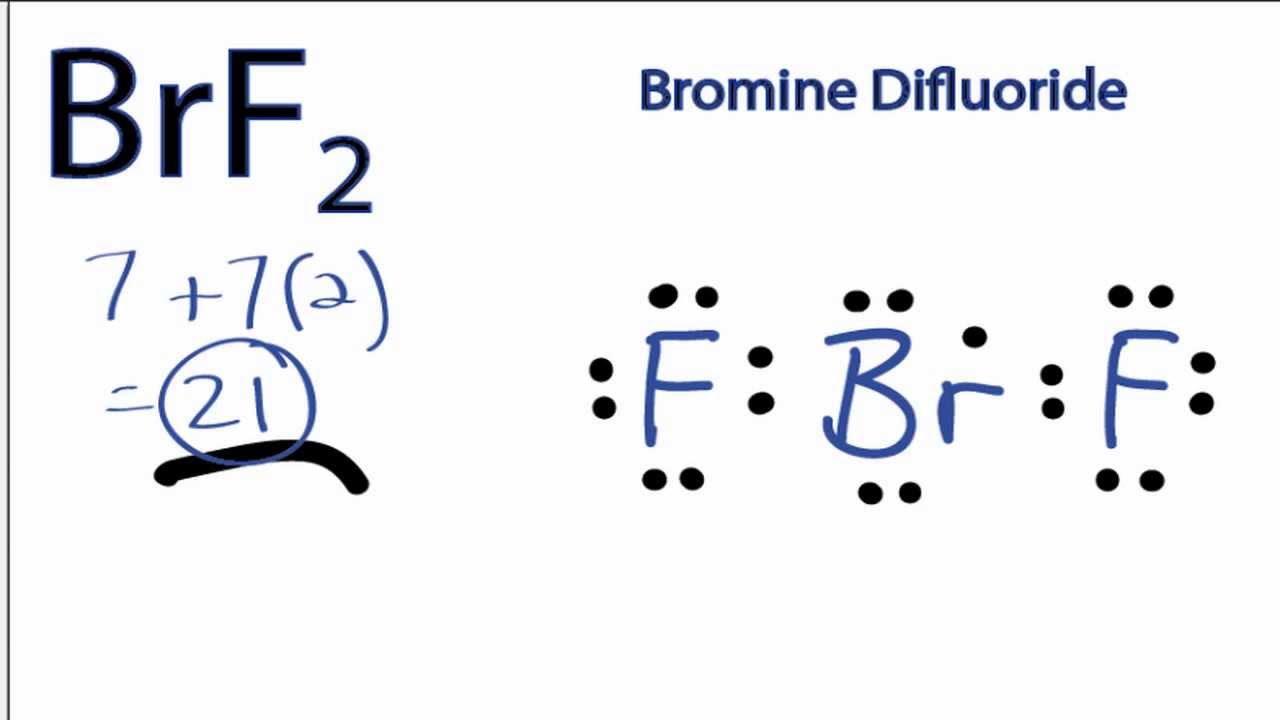
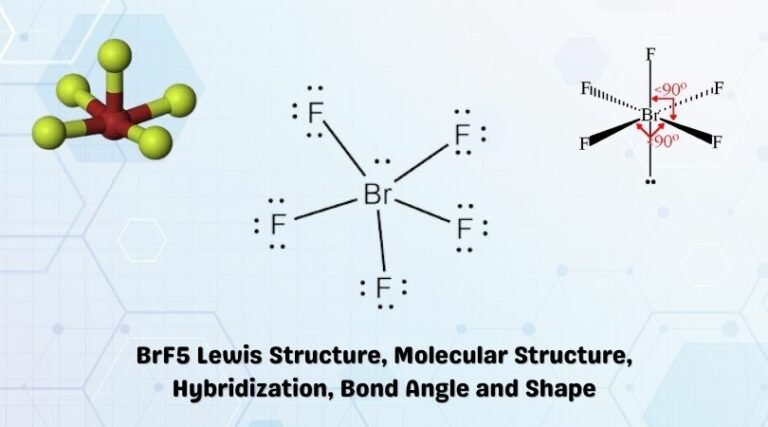
Where two or more resonance structures can represent a molecule,the VSEPR model is applicable to any such structure. A multiple bond is treated as if it is a single electron pair and the two or three electron pairs of a multiple bond are treated as a single super pair. The valence shell is taken as a sphere with the electron pairs localizing on the spherical surface at maximum distance from one another. These pair of electrons tend to occupy such positions that minimize repulsion and thus maximize distance between them. Decision: The molecular geometry of BrF5 is square pyramidal with asymmetric charge distribution on the central atom. Pairs of electrons in the valence shell repel one another since their electron clouds are negatively charged. The square pyramidal effect of BrF5 molecular structures is. These + and - charges are responsible to make the entire BrF5 molecule polar. The shape of a molecule depends upon the number of valence shell electron pairs (bonded or non-bonded) around the central atom. It indicates that the basic atom has a single pair of electrons. BrF5 is a POLAR molecule because the Br-F bonds present in the molecule are polar and it has asymmetric geometry which causes the partial positive (+) and partial negative (-) charge to appear on the molecule. The molecular geometry of BrF5 is square pyramidal with an asymmetric. Now we discuss the main postulates of VSEPR theory are as follows: Pbr5 Molecular Geometry Lewis Structure Shape Bond Angle And More ( jpg). Hence, we can easily find the hybridization of IF5 using these two methods. Hence, H1/2 7+5 H6, indicating that its Sp3d2 hybridized. In the case of IF5, V 7 (valence electrons of central atom) M 5 (5 monovalent atoms of F) Since the total charge of IF5 is 0, C and A will be zero. Hint:To be able to predict the shape of molecules,it is important to learn about the Valence Shell Electron Pair Repulsion Theory,also known as the VSEPR Theory.This theory provides a simple procedure to predict the shape of molecules depending on various factors such as presence of lone pairs around the central atom, number of bonding pairs, repulsive interactions of electron pairs and many more which will be discussed. H6, the molecule will have Sp3d2 hybridization.
